Final results from landmark DYNAGITO® trial published in The Lancet Respiratory Medicine[1]
Results enhance existing evidence base showing Spiolto® Respimat® improves symptom reduction and quality of life over Spiriva® Respimat®[2,3,4,5,6]
For media excluding the United States of America, Canada and the United Kingdom
Final results from landmark DYNAGITO® trial published in The Lancet Respiratory Medicine
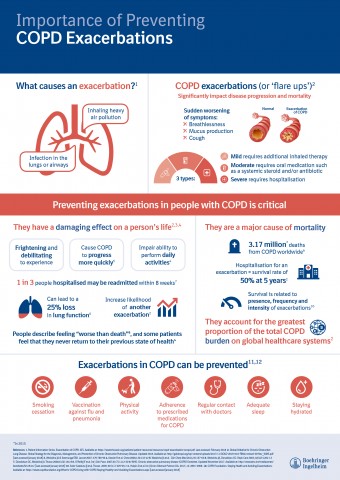
COPD is a progressive, yet treatable condition that significantly impacts patients’ lives, restricting their daily activities from early on in the disease.[8,9,10] COPD exacerbations, or flare-ups, are sudden episodes of increased breathlessness, cough and mucus production that can last for several days or even weeks.[11] These episodes can be seriously disabling, resulting in a need for urgent medical care, including hospitalisation, and sometimes lead to death.[11]
Further DYNAGITO® data demonstrated that tiotropium/olodaterol was associated with fewer moderate-to-severe exacerbations that needed intervention with a systemic corticosteroid, with or without antibiotics, compared with tiotropium:[1*]
A 20% lower rate of moderate-to-severe exacerbations that required treatment with a systemic corticosteroid (p=0.0068).[1*]
A 9% lower rate of exacerbations where the use of both a systemic corticosteroid and antibiotics were needed (p=0.0447).[1*]
No difference was observed in the rate of exacerbations that required treatment with antibiotics only (p=0.2062).[1*]
No new side effects or safety concerns were identified in the DYNAGITO® study.[1] These data also show that tiotropium/olodaterol has a similar safety profile to tiotropium.[1]
Reducing symptoms and the future risk of exacerbations are key treatment goals for COPD.[7] According to the international GOLD† 2018 Strategy recommendations, LAMA/LABA treatments such as tiotropium/olodaterol play a central role in the management of COPD and help to achieve these treatment goals.[7]
Intended audiences:
This press release is issued from our Corporate Headquarters in Ingelheim, Germany and is intended to provide information about our global business. Please be aware that information relating to the approval status and labels of approved products may vary from country to country, and a country-specific press release on this topic may have been issued in the countries where we do business.
For references and notes to editors, please visit: http://www.boehringer-ingelheim.com/press-release/results-landmark-dynagito-trial
* The pre-specified significance level of p<0.01 for the primary endpoint of DYNAGITO® was not met
† Global Initiative for Chronic Obstructive Lung Disease
View source version on businesswire.com: https://www.businesswire.com/news/home/20180329005455/en/
Website: http://www.boehringer-ingelheim.com
Contact
Boehringer Ingelheim
Corporate Communications
Media + PR
Dr. Carolin Grob
carolin.grob@boehringer-ingelheim.com
Phone: +49 (6132) 77-182603
Fax: +49 (6132) 77-6601
press@boehringer-ingelheim.com
This news is a press release provided by Boehringer Ingelheim International GmbH.